Abstract
Off-the-shelf CAR T cells potentially offer advantages over autologous strategies such as ease of manufacturing, quality control, avoidance of malignant contamination and T cell dysfunction, as well as the ability to generate a more consistent CAR T product from healthy T cells. However, the vigorous host-versus-graft immune response against histoincompatible T cells prevents expansion and persistence of allogeneic CAR T cells and mitigates the efficacy of this approach. A major challenge is that, while HLA deletion can result in adaptive immune evasion, innate reactivity is enhanced with this approach. CD47 overexpression can block both NK cell and macrophage killing (J Exp Med 2021;218(3):e20200839), and we hypothesized that T cells would lose their immunogenicity when human leukocyte antigen (HLA) class I and II genes are disrupted and CD47 is over-expressed. We describe here the engineering of human immune evasive CAR T cells building on our previously described hypoimmune technology (Nat Biotechnol 2019;37(3):252-258 and Proc Natl Acad Sci U S A 2021;118(28):e2022091118).
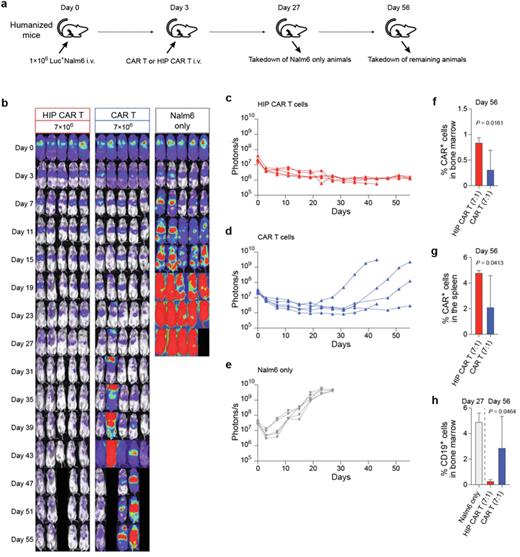
Human T cells from healthy donors were obtained by leukapheresis. CRISPR/Cas12b technology was used to disrupt the B2M, CIITA, and TCR genes, and lentiviral transduction was used to overexpress CD47 and to express a CD19 CAR to generate hypoimmune (HIP) CD19 CAR T cells. Control T cells were unmanipulated except for overexpression of the CD19 CAR. When transplanted into allogeneic humanized mice, hypoimmune CAR T cells evade immune recognition by T cells using ELISPOT analysis. In contrast, transplantation of control CD19 CAR T cells generated from the same human donor resulted in a significant T cell activation (p<0.0001 unpaired t-test). Innate immune cell assays show that CD47 overexpression protects HLA-I/II deficient CD19 CAR T cells from NK cell and macrophage killing in vitro and blocking of CD47 results in killing by innate allogeneic immune cells.
For persistence studies, allogeneic humanized mice were injected with 1´106 Luc+ Nalm6 cells and received 7´106 control CD19 CAR T cells or HIP CD19 CAR T cells. However, CD19 CAR T cells are recognized and rejected by the allogeneic immune system of the humanized mice, leading to tumor grow in this group. Flow cytometry on Day 56 from bone marrow and spleen confirms persistence of HIP CD19 CAR T cells, but not control CD19 CAR T cells (see Figure). These findings show that HIP CD19 CAR T cells are functionally immune evasive in allogeneic recipients with cytotoxic anti-tumor capacity and suggest they could provide universal HIP CAR T cells that are able to persist without immunosuppression. Furthermore, these data suggest that HIP CD19 CAR T cells can be used for re-dosing strategies.
Disclosures
Hu:Sana Biotechnology Inc: Current Employment, Current equity holder in publicly-traded company. Manner:Sana Biotechnology Inc: Current Employment, Current equity holder in publicly-traded company. Gattis:Sana Biotechnology Inc: Current Employment, Current equity holder in publicly-traded company. Ngo:Sana Biotechnology Inc: Current Employment, Current equity holder in publicly-traded company. De Jesus:Sana Biotechnology Inc: Current Employment, Current equity holder in publicly-traded company. Young:Sana Biotechnology Inc: Current Employment, Current equity holder in publicly-traded company. Wells:Sana Biotechnology Inc: Current Employment, Current equity holder in publicly-traded company. Weng:Sana Biotechnology Inc: Current Employment, Current equity holder in publicly-traded company. White:Sana Biotechnology Inc: Current Employment, Current equity holder in publicly-traded company. Dowdle:Sana Biotechnology Inc: Current Employment, Current equity holder in publicly-traded company. Foster:Sana Biotechnology Inc: Current Employment, Current equity holder in publicly-traded company. Fry:Sana Biotechnology Inc: Current Employment, Current equity holder in publicly-traded company. Schrepfer:Sana Biotechnology Inc: Current Employment, Current equity holder in publicly-traded company.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal